
Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
Ru(II)-amine ligand based complexes developed by Noyori and co-workers are very active and selective catalysts for the hydrogenation or asymmetric hydrogenation of ketones. This achiral ligand example illustrates how the hydrogen is transferred to the carbonyl.
Click for an activated asymmetric enantioselective hydrogenation reaction
Further Information:
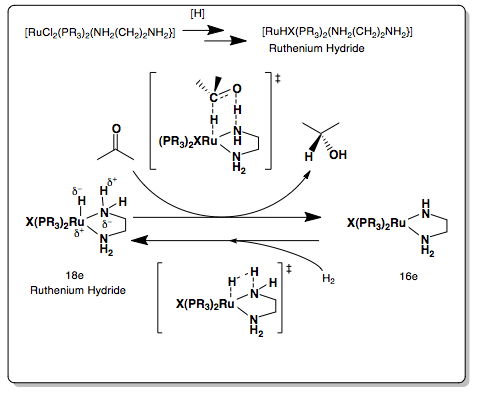
The Ruthenium hydride is formed by adding an alkaline base and a hydride source. When the carbonyl is added hydrogen is transferred from the ruthenium hydride ligand to the carbonyl. There is no interaction of the ruthenium with the carbonyl oxygen during the hydrogenation. Two electrons are lost as the two hydrogen are transferred to the carbonyl to form the propan-2-ol in a pericyclic six-membered transition state. The ruthenium hydride can be produced again with addition of H2.
R. Noyori and T. Ohkuma, Angew. Chemie – Int. Ed., 2001, 40, 40–73.