NOTE: Important charges and non-bonding electrons are shown throughout the animation except during the transition phase
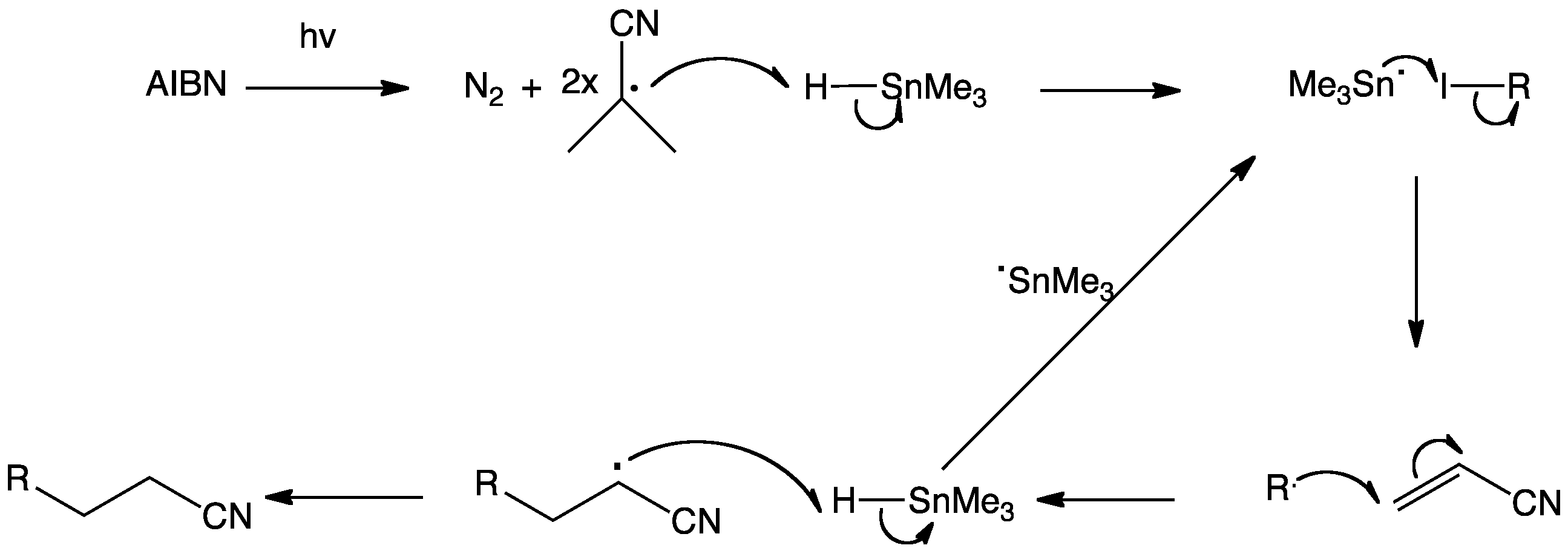
The chain is initiated through the homolysis of azobis(isobutyronitrile) (AIBN) to two nitrile-stabilised radicals and nitrogen gas. Abstraction of iodine by the tin radical produces an alkyl radical which can undergo conjugate addition to acrylonitrile. The resulting electrophilic radical then abstracts hydrogen from trimethyltin hydride. The resulting trimethyltin radical is then recycled to produce more of the alkyl radical.
Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
K. Miura, K. Ootsuka and A. Hosomi, Synlett, 2005, 0, 3151–3153.