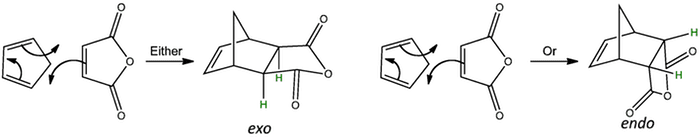
Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
In this case both the diene and dienophile have stereochemistry. Although there are four stereogenic centres in the product only two diastereoisomers can be formed, any others are impossibly strained. The diastereoisomers formed are known as exo and endo.
The exo transition state looks like the letter ‘Z’ on it’s side, whereas the endo transition state looks like the letter ‘U’. Rotate the transition states to see the difference.
The exo product is most stable as the smaller one-atom bridge eclipses the anhydride ring causing less steric hindrance, and is the thermodynamic product. Under reversible conditions, the exo product is formed. However, under normal conditions, the less stable endo product is formed and is the kinetic product of the reaction.