Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
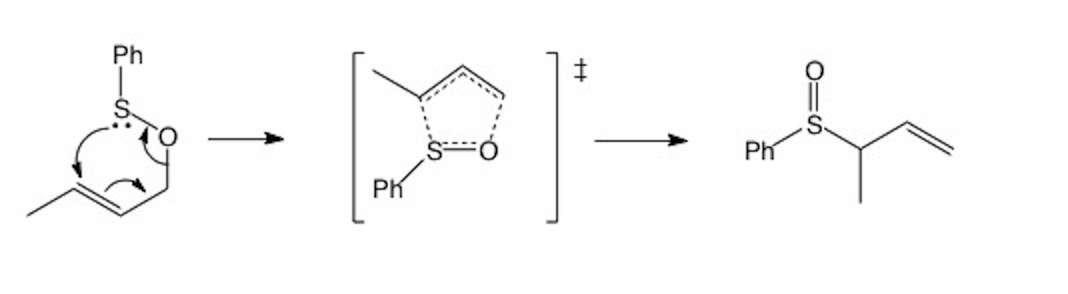
All [3,3]-sigmatropic rearrangements have six-membered cyclic transition states. As with all reactions in this class, the sum of the two numbers in the brackets gives the size of the ring in the transition state. This means we will see five-membered rings in the transition states of [2,3]-sigmatropic rearrangements. Can you count round the reactant, from the old to the new sigma bond position and verify the (2,3) relationship? (Hint: include in your count the atom forming the sigma bond). Notice the chair-like part of the five-membered transition state.
Richard Windsor – Undergraduate Final Year Project 2008
D. A. Evans and G. C. Andrews, Acc. Chem. Res., 1974, 7, 147–155.