The Swern oxidation is a modern method of converting primary or secondary alcohols to aldehydes or ketones using oxalyl chloride, dimethyl sulfoxide (DMSO) and a base (Et3N) at low temperature. The overall reaction is as follows:
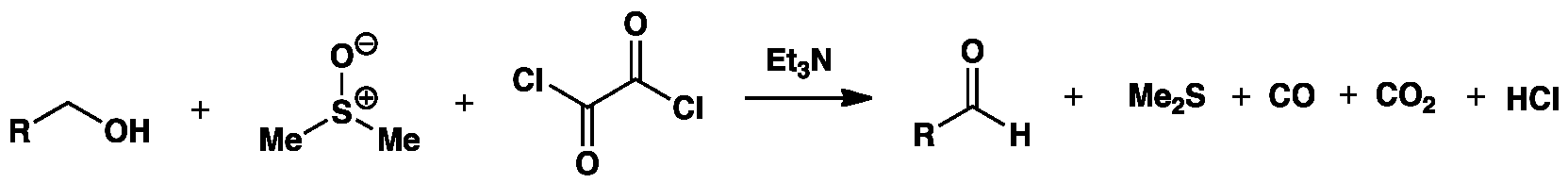
The three stages of the Swern oxidation reaction mechanism are shown below.
Click the different stages to view the 3D models of the reaction