Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
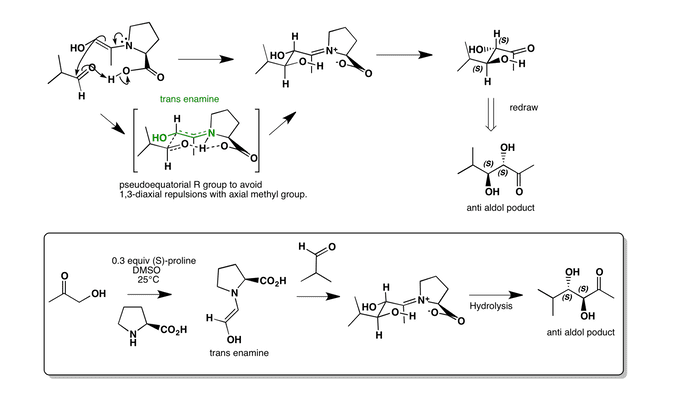
Asymmetric intermolecular aldol catalysed by S-proline. The reaction goes via a hydroxyacetone enamine, with the -OH group trans to the proline. It attacks the aldehyde on it’s re-facial side to form and anti product. In the chair like transition state the isopropane group is pseudoequatorial to minimise the 1,3-diaxial repulsions from the axial methyl group alpha to the nitrogen. This forms the (S,S)-aldol product, which is anti.
W. Notz and B. List, J. Am. Chem. Soc., 2000, 122, 7386–7387.