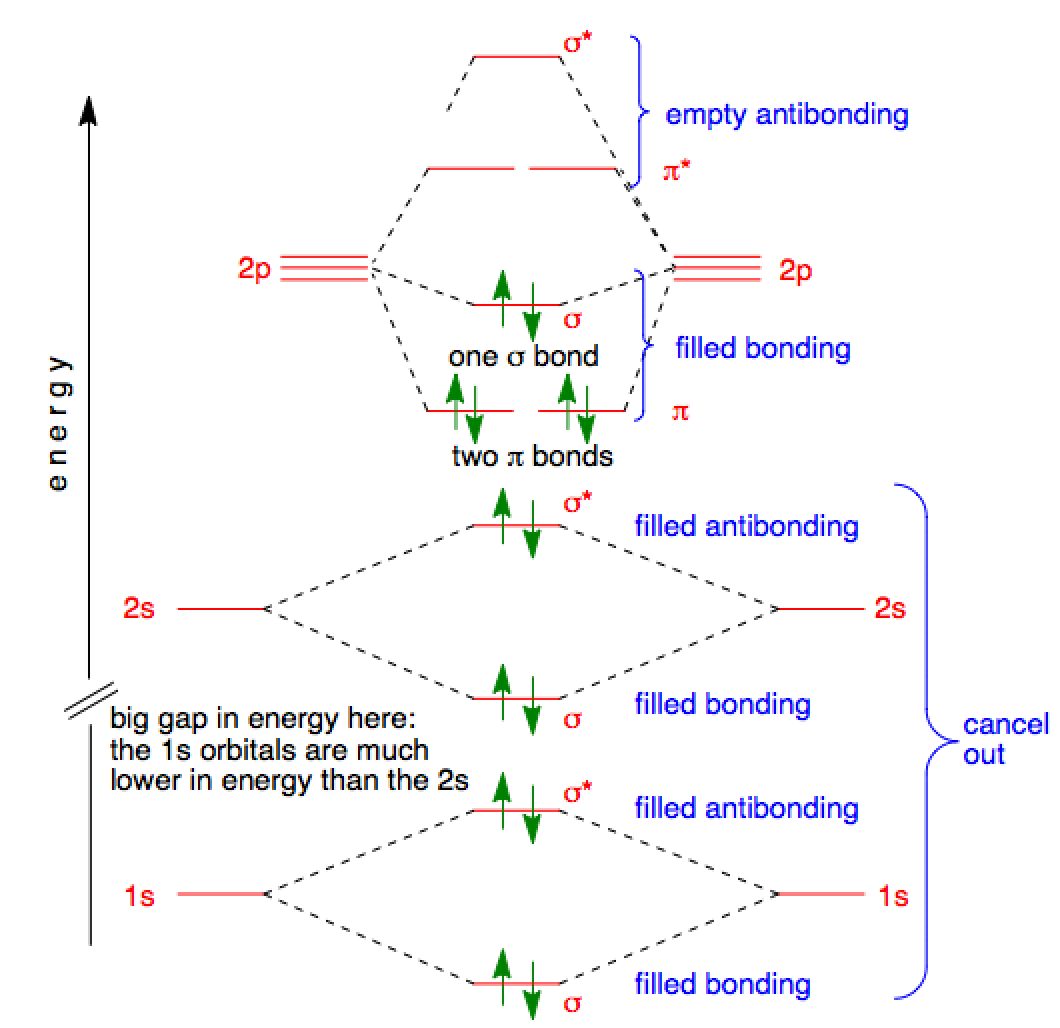
There are four molecular orbitals derived from the 1s and 2s orbitals.
Use the buttons to display the 1s and 2p atomic orbitals that make up the molecular orbitals.
The p orbitals combine to produce a sigma and two perpendicular pi bonds. Three filled bonding orbitals…
… and three empty antibonding orbitals.
The molecular orbital energy-level diagram for Period 2 homonuclear diatomic molecules from Li2 to N2.
Explore bonding orbitals in other small molecules
Hydrogen | Fluorine | Nitrogen | Hydrogen Fluoride | Carbon Monoxide | Methane | Ammonia | Ethylene | Acetylene | Allene | Formaldehyde | Benzene