Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
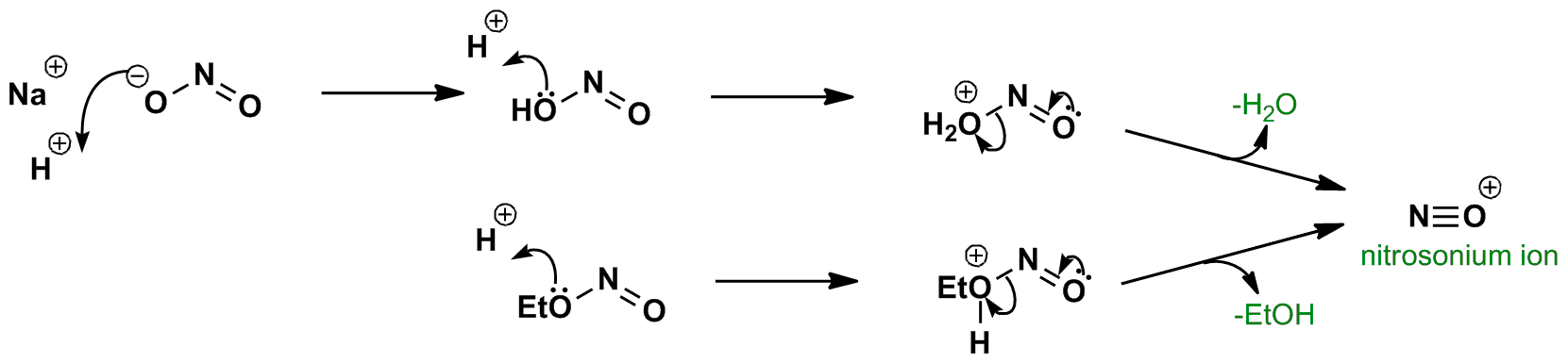
Sodium nitrite and strong acid e.g. HCl produce the weak acid nitrous acid which is further protonated and loses water to generate the nitrosonium ion. It can also be formed from alkyl nitrites in the presence of acid. The NO+ nitrosonium ion is used in the formation of diazonium salts.
F. Mo, G. Dong, Y. Zhang and J. Wang, Org. Biomol. Chem., 2013, 11, 1582.