Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
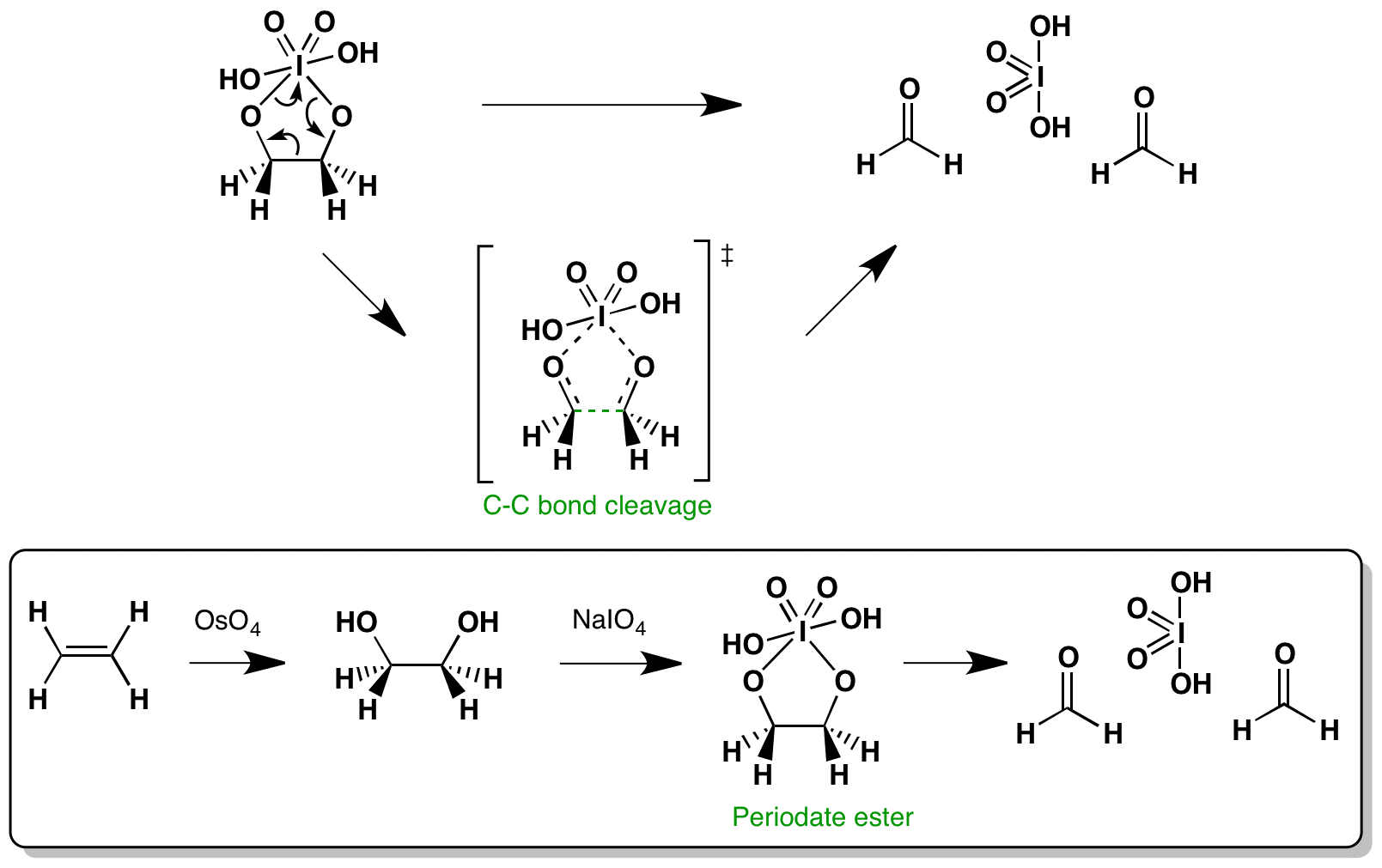
1,2-Diols can be cleaved oxidatively with sodium periodate NaIO4 (and lead tetraacetate) to form two carbonyl compounds in this case formaldehyde, and periodic acid. This proceeds via a cyclic transition state.
When preceeded by dihydroxylation with osmium tetroxide, the sequence is an alternative to ozonolysis of alkenes.