Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
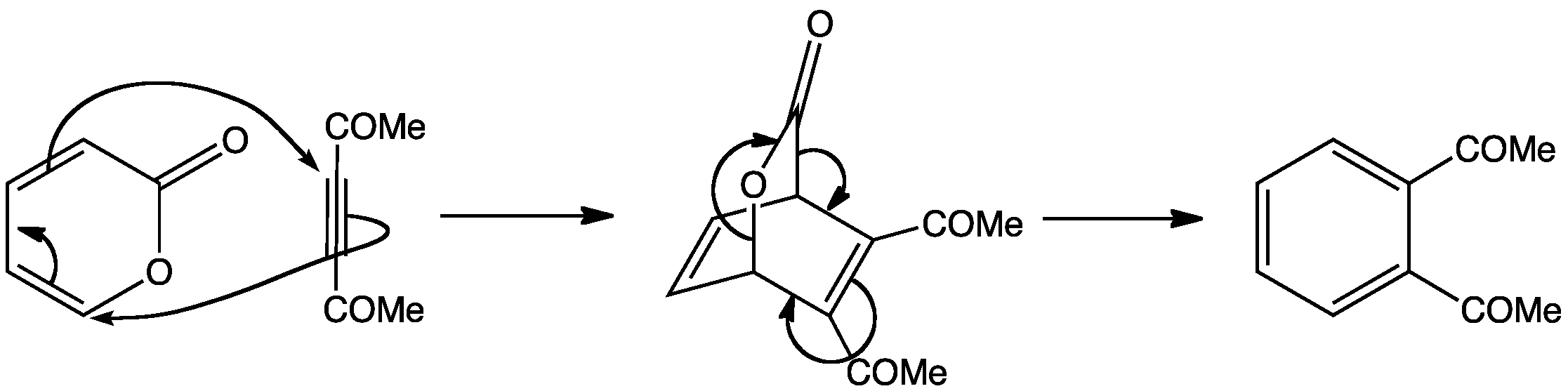
Pyrones can be used as routes to aromatic rings, however, they are rather unstable and barely aromatic. They can react with alkynes via a Diels-Alder reaction, followed by a reverse Diels-Alder reaction, involving the loss of carbon dioxide, to produce benzene derivatives.
K. Afarinkia, V. Vinader, T. D. Nelson and G. H. Posner, Tetrahedron, 1992, 48, 9111–9171.