Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
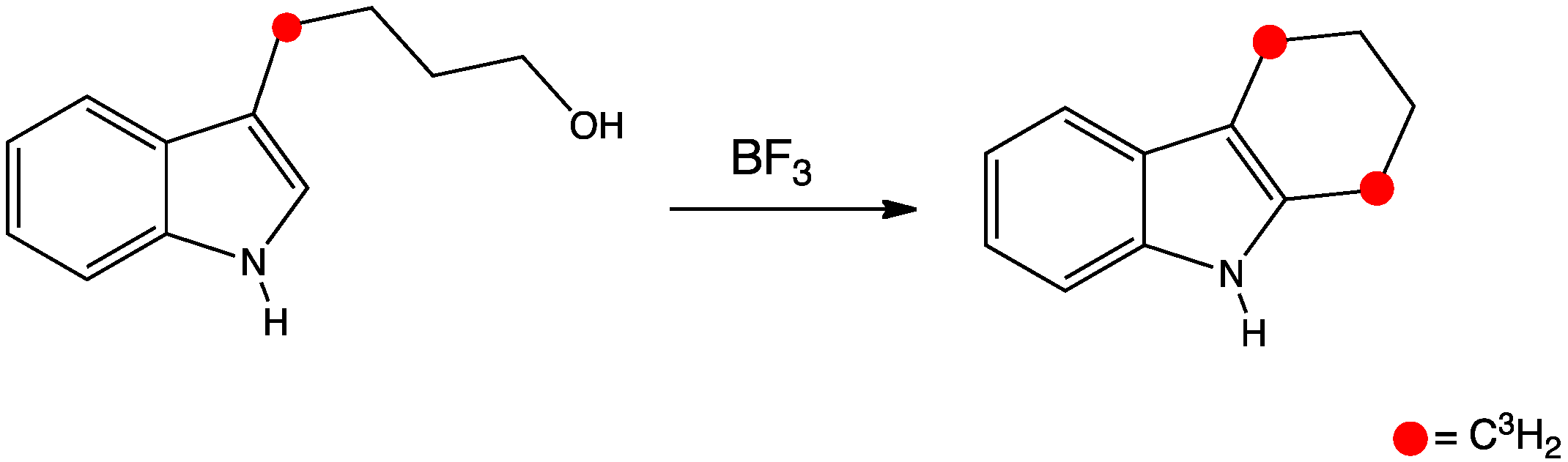
This reactions shows a Friedel-Crafts type cyclisation. However, it is not quite as simple as it first seems! An experiment was divised, where the starting material was labelled with tritium (radioactive 3H) as shown in the diagram below. The product then shows 50% of the label where it is expected and 50% where it is not. This result suggests a symmetrical intermediate.
The product is formed from a intermediate spiro compound, which has a 5 membered ring at right angles to the indole ring so each CH2 group has an equal chance to migrate.
This mechanism shows typical indole reactivity, where is reacts through the 3-position to maintain the aromaticity of the benzene ring.