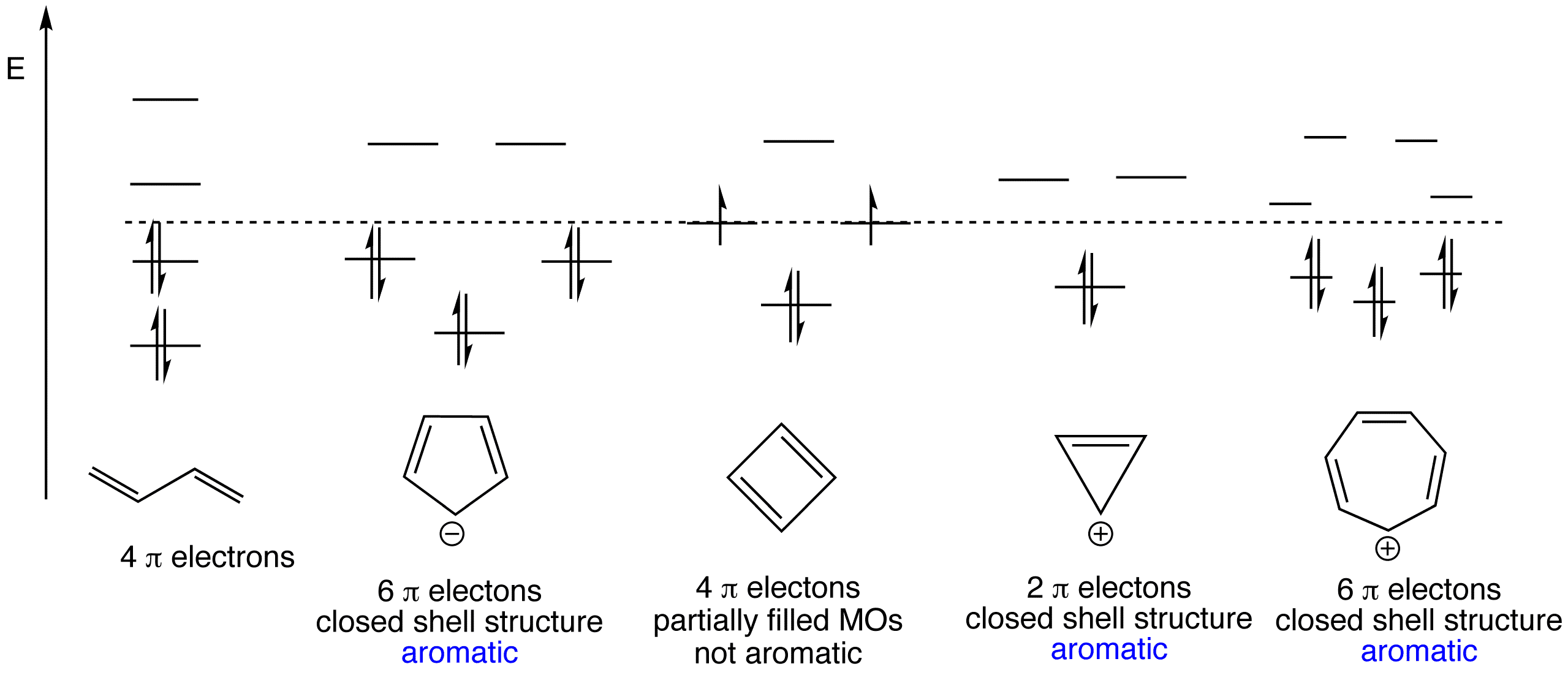
Click the structures in turn and then the orbital energy levels to view the 3D models and molecular orbitals respectively.
Butadiene, cyclopentadienyl anion, cyclobutadiene, cyclopropylium cation and tropylium cation are featured here.
The calculated energy of the molecular orbital is shown in the top left corner.
• You can observe the degeneracy of pairs of molecular orbitals.
• Notice that increasing the number of nodes corresponds to increasing energy.