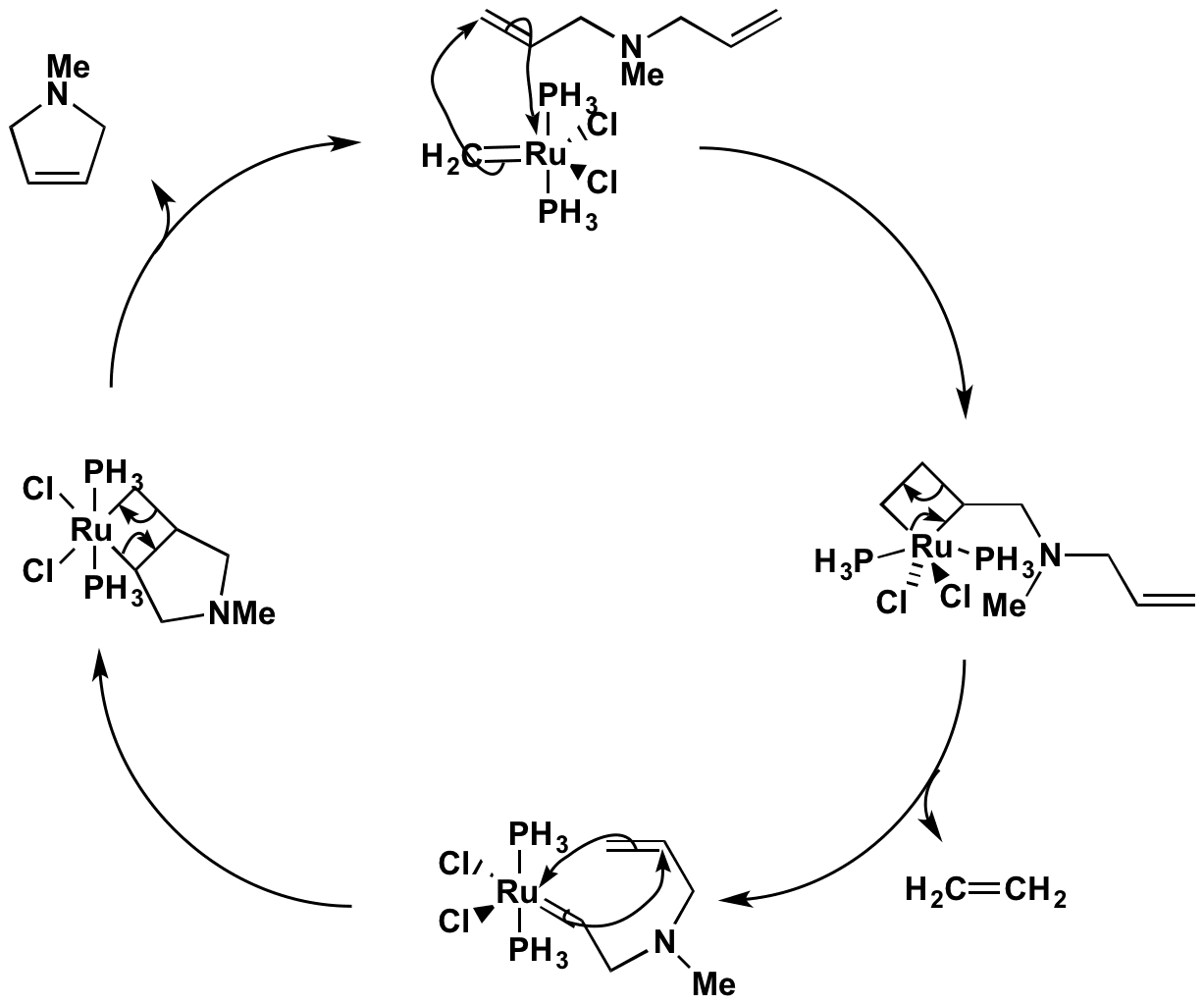
Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
The ruthenium carbene complex catalyst is reformed at the end of the reaction (usually P(cyclohexyl)3 ligands are used instead of PH3), making the process very efficient. The apparent selectivity in the first part is due to entropy – the release of ethene gas. In the second part, if the Ru adds to the other end of the alkene, then the intermediate is symmetrical and can decompose only to the starting material.
For another olefin metathesis reaction closing a 12-membered ring.
G. C. Vougioukalakis and R. H. Grubbs, Chem. Rev., 2010, 110, 1746–1787.
T. J. Donohoe, J. F. Bower and L. K. M. Chan, Org. Biomol. Chem., 2012, 10, 1322.
A. V. Gulevich, A. S. Dudnik, N. Chernyak and V. Gevorgyan, Chem. Rev., 2013, 113, 3084–3213.