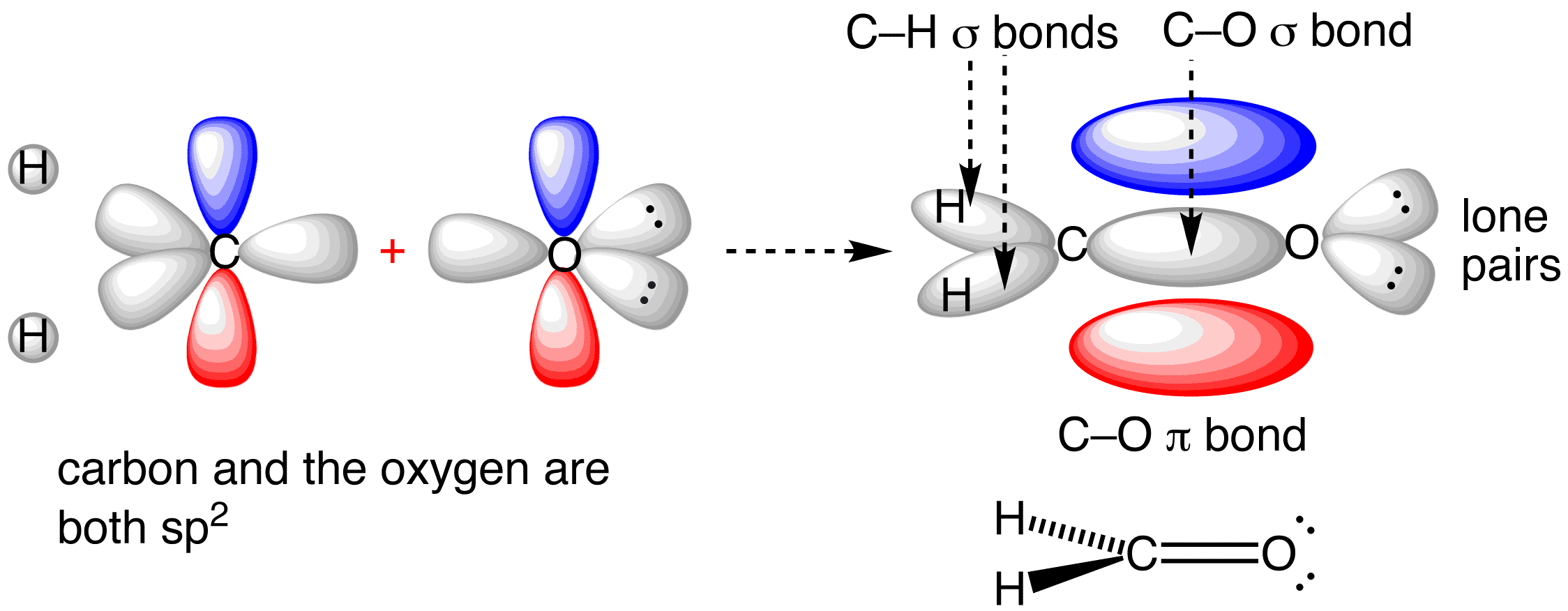
Use the buttons to display the sp2 orbitals that make up the sigma framework and the remaining p orbitals which form the pi-bond.
Explore bonding orbitals in other small molecules
Hydrogen | Fluorine | Nitrogen | Hydrogen Fluoride | Carbon Monoxide | Methane | Ammonia | Ethylene | Acetylene | Allene | Formaldehyde | Benzene