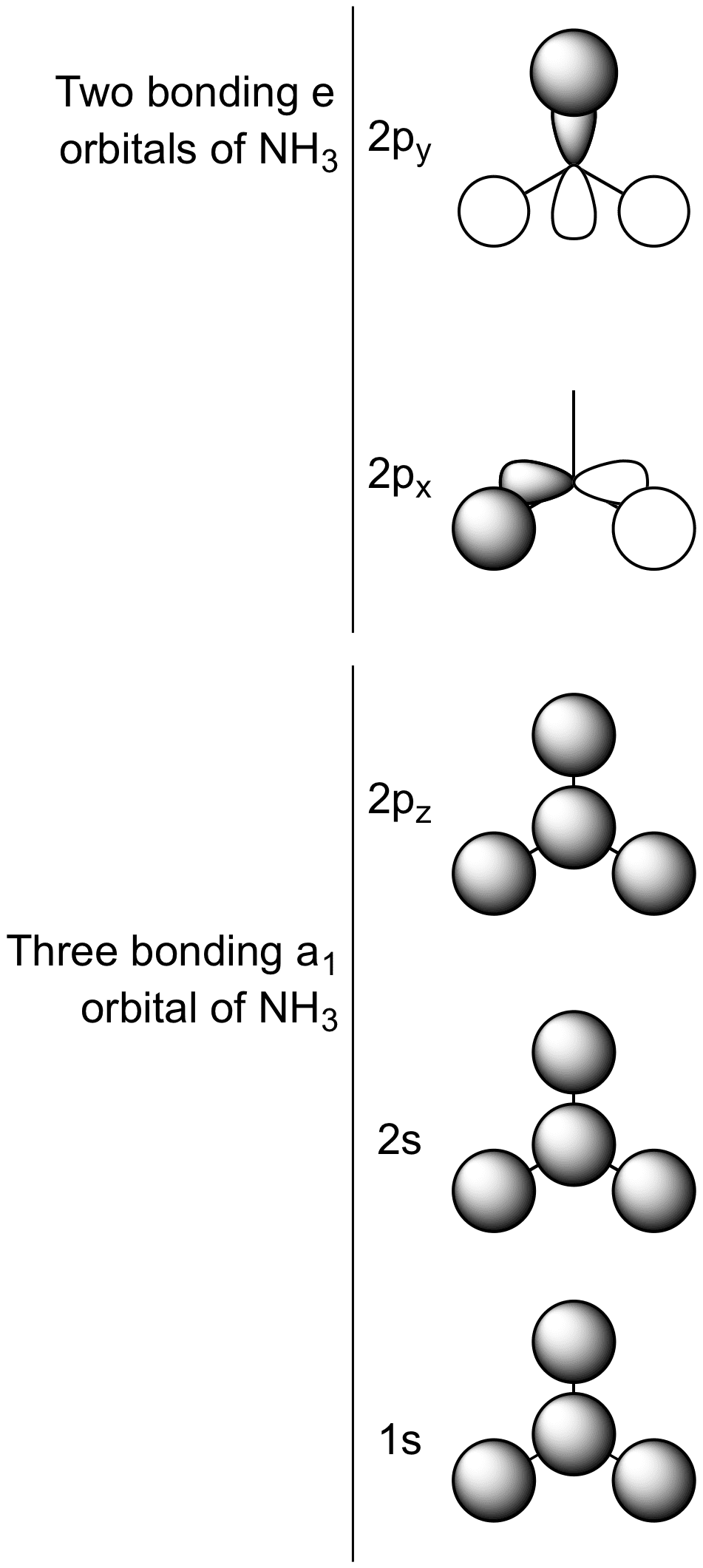
Specific combinations of atomic orbitals are use to build molecular orbitals.
As seen here the pz orbital, like the 1s and 2s orbitals, has A1 symetry; they are totally symmetric to the operations of the C3v point group.
The px and py are affected by the C3v point group operations by equal amounts and hence are degenerate. These atomic orbitals have e symmetry as defined by the character table.
Hence a combination of the appropriate atomic orbital on Nitrogen with the appropriate Symmetry Adapted Linear Combination (SALC) of hydrogen 1s orbitals generates the molecular orbitals shown above.
SALC – Ammonia Atomic Orbitals
Orbital-orbital overlap and SALC Homepage