NOTE: Important charges and non-bonding electrons are shown throughout the animation except during the transition phase
Click the structures and reaction arrows to view the 3D models and animations respectively
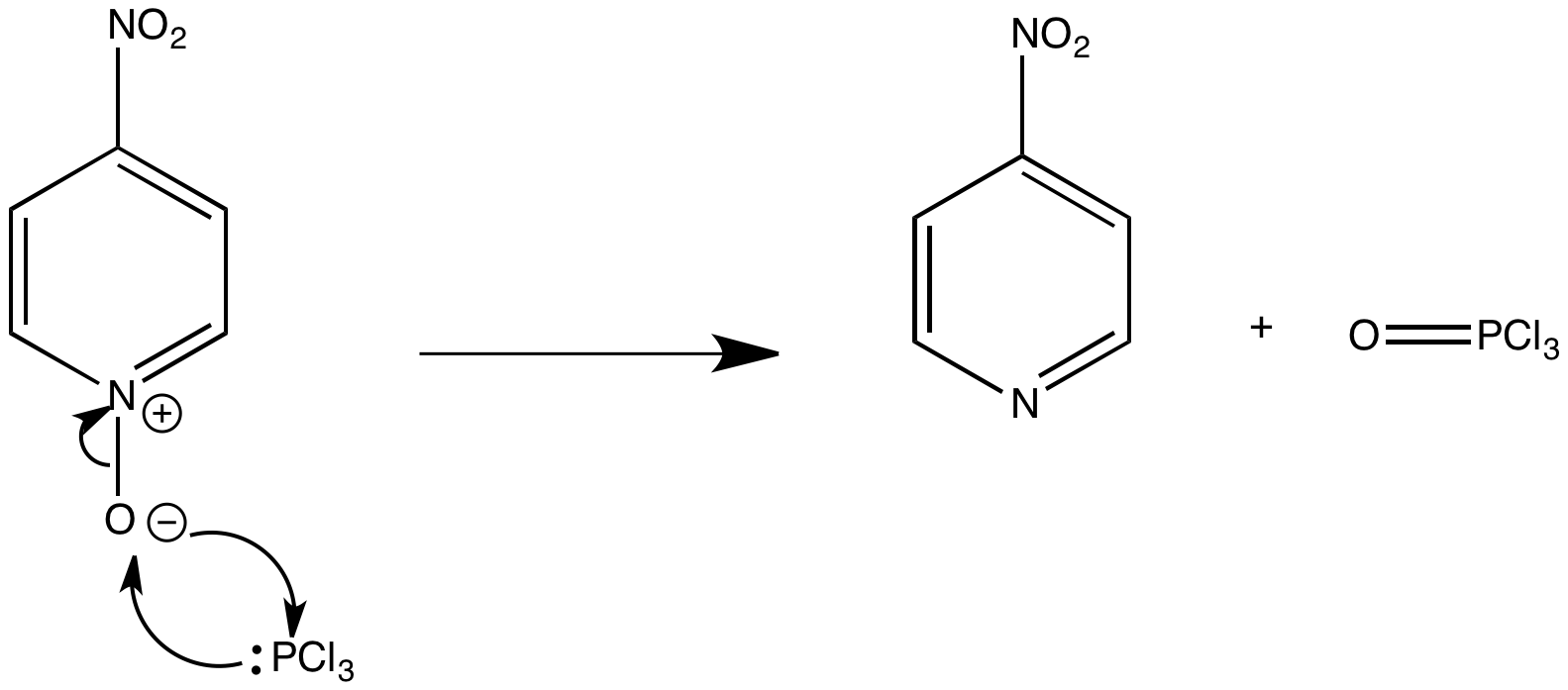
Cleavage of the N-oxide bond usually occurs using trivalent phosphorus compounds. The phosphorus donates its lone pair of electrons whilst accepting a pair of electrons into a d orbital. The formation of the strong P=O bond is a driving force for the reaction.