Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
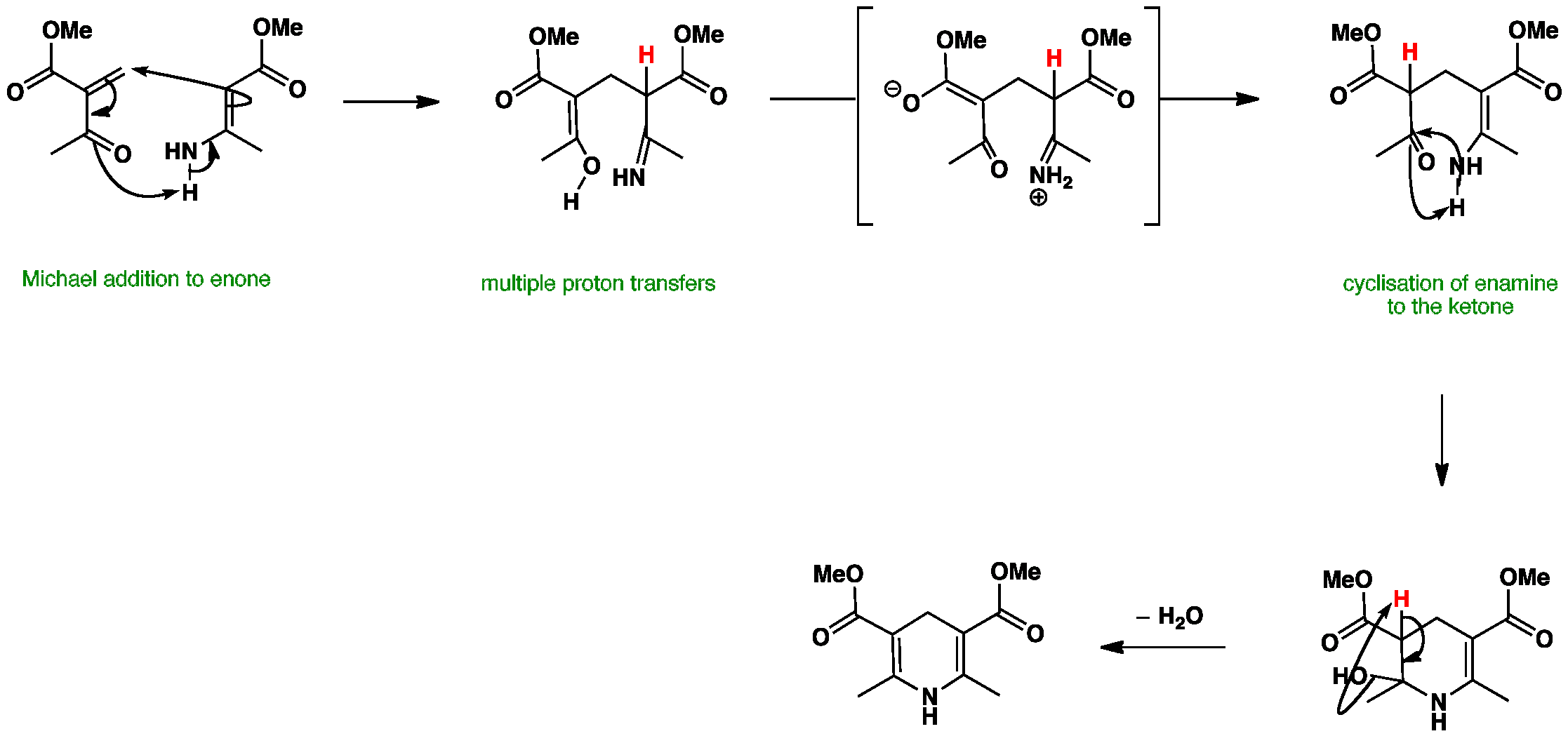
A conjugate addition between the two fragments, followed by proton transfers and finally intramolecular enamine formation gives the dihydropyridine. One of the possible sequences is shown here.
H. G. O. Alvim, E. N. da Silva Júnior and B. A. D. Neto, RSC Adv., 2014, 4, 54282–54299.