Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
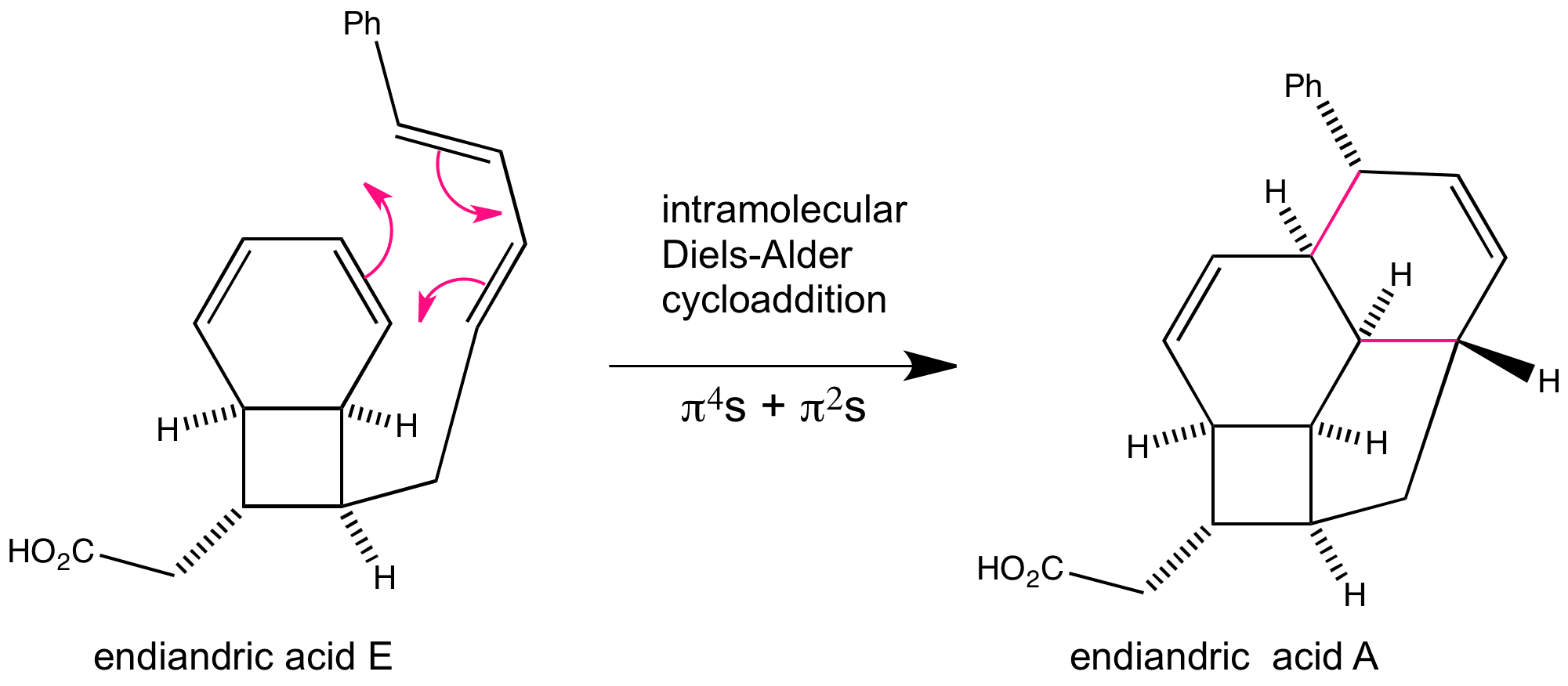
This is an example of an intramolecular Diels-Alder reaction that occurs in Nature where the diene and dienophile are contained within the same molecule, endiandric acid E.
In this sequence of tandem organic reactions, the previous series of electrocyclic reactions have set up the system to be able to carry out another pericyclic reaction, cycloaddition.
Final Year Project 2013: Ilona Blee
K. C. Nicolaou, N. A. Petasis, R. E. Zipkin and J. Uenishi, J. Am. Chem. Soc., 1982, 104, 5555–5557.
961