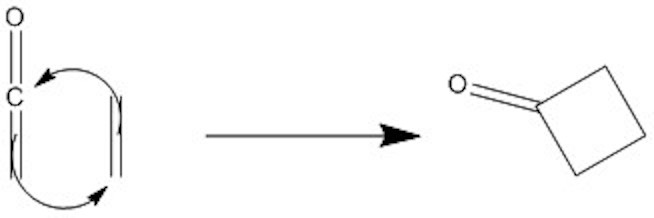
Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
[2+2] Ketene Cycloadditions are one of the few [2+2] thermal cycloadditions allowed.
The [2+2] Ketene addition is allowed as the transition state is stablised by the pi orbital of the Carbonyl stablising the electron difficent carbon on the alkene. This is shown by the CO MO stablisation button above.
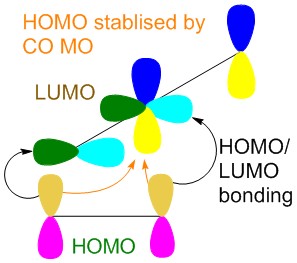
The diagram above shows how the carbonyl orbital helps to stablise the alkene.