Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
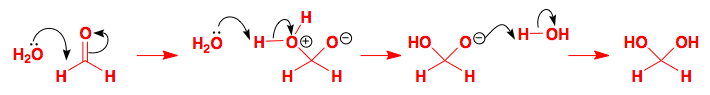
Formaldehyde is an extremely reactive aldehyde as it has no substituents to hinder attack—it is so reactive that it is rather prone to polymerization. An aqueous solution of formaldehyde contains essentially no CH2O—it is completely hydrated.
Nucleophilic addition of water to the reactive carbonyl group is followed by proton transfers from one oxygen atom to the other mediated by water molecules.