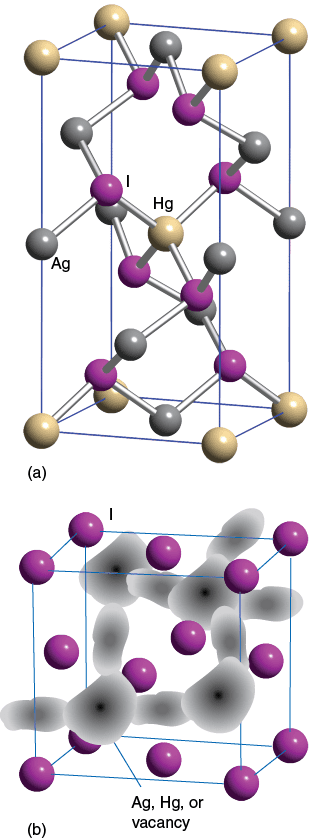
In its low temperature form, Ag2HgI4 adopts an ordered structure (a) in which the Ag+ and Hg2+ ions occupy some of the tetrahedral sites in the structure. At high temperatures, structure (b) is adopted, there are more sites in the structure than there are ions (Ag+ and Hg2+) present. As a result, the conductivity increases as the ions are more mobile and the I– ions are easily polarizable.