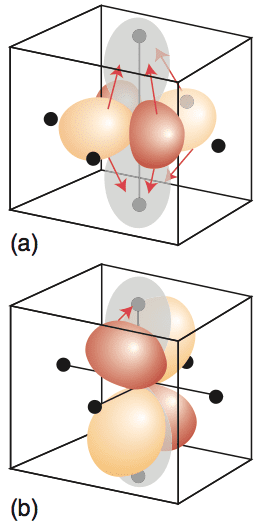
Click the top orbital image to load the orbitals, then click below for the second combination.
Electron movement is symbolised by shading of the orbitals.
Shifts in electron density that accompany the two transitions occur. There is a considerable relocation of electron density towards the ligands on the z-axis in (a), but a much less substantial relocation in (b).